Where Can You Find Phentermine for Sale? See Below:

Phentermine 37.5
The drug Phentermine 37.5 mg is the most popular dosage because it is extremely potent and prescribed by many doctors. The drug is used to treat obesity in patients by suppressing appetite… Learn More See Pricing

Adipex
Adipex is the trade name for medication that contains phentermine.Adipex must be taken only upon prescription and is helpful for weight loss among people who regularly exercise and follow a healthy diet plan. Learn MoreSee Pricing

Ionamin
A brand name of phentermine, Ionamin, is a hunger suppressant which is used alongside diet and exercise for obesity management. It is among the earliest drugs meant to beat obesity and got approval from FDA over 50 years ago… Learn MoreSee Pricing

Qsymia
One of the solutions of losing extra fat could be Qsymia, which is a drug that containing a combination of topiramate and phentermine. The drug was approved as an extended-release form by the US FDA in 2012. It can be consumed by adults who have a BMI of 30 or mor… Learn MoreSee Pricing
Phentermine Timeline
1950: Phentermine Gains Regocnition: The early 1950s saw phentermine being touted as the first hunger suppressant in the US market. The drug was marketed as an extremely potent weight-loss medicine that fought obesity because of its suppressant nature.
1959: Phentermine Gets FDA Approved: increasing market share of the drug allowed it to gain mainstream popularity and phentermine was finally approved by the US Food and Drug Administration (FDA) in 1959.
1970: Phentermine hydrochloride Introduced: 1970s saw the release of phentermine hydrochloride, which could be used over-the-counter as well. Later, in 1984, the medicine was combined with another drug, Fenfluramine
2012: Qsymia Introduced: An evolved form of phentermine, the “Qsymia” was rejected by FDA in 2010. Two years later however, (February 2012), the federal body voted in favor of Qsymia and later (July 2012), approved it for sale in the US consumer market.
Phentermine: An infamous weight loss drug
Phentermine is a drug that has a notorious history around the world. Phentermine is chemically an amphetamine and it is basically a short form for phenyl-tertiary-butylamine. Being an amine, phentermine is formally a derivative of ammonia and is considered to be a tertiary amine. The drug can be produced by synthesizing 2-nitropropane and benzaldehyde.
Medical researchers were initially interested in how amphetamines affected the bodies of soldiers during the Second World War. Amphetamines were used to increase concentration levels and mental alertness in soldiers, while they decreased symptoms of fatigue and hunger. This is what primarily led to researchers in the United States studying the effects of amphetamines after the conclusion of the war. Corporations also saw potentially huge markets if drugs based on amphetamines were made available to the general public.
Since 2012, the FDA has approved around four weight loss products, but phentermine has continued to be remain the market leader in this segment. As long as the price of phentermine remains relatively low when compared to its competition, the drug will continue to be the preeminent drug in the market.
A brief history of phentermine
In the United States, phentermine was approved by the Food and Drug Administration (FDA) in 1959 and was made available for commercial sales in the same year. The first version of this drug was phentermine resin and the FDA approved its usage for the short-term treatment of obesity. Phentermine was one of the first commercially available appetite suppressants in the United States. Due to yet understood and potentially dangerous side effects, the drug was limited to a ninety day administration period when it was released to the public in 1959. The FDA wanted more research to be carried out on the drug before it was prescribed to patients for an indefinite period of time.
Phentermine managed to build up its reputation as a generic drug that effectively combated obesity. In the 1970s, the drug was sold as phentermine hydrochloride with HCL (hydrochloric acid) becoming a part of the drug’s chemical composition. When it was released in this form, it did not have any limited administration period.
Phentermine hydrochloride is typically sold in the form of 37.5 mg tablets. The effect of this is equivalent to about 30mg of pure phentermine. The normal adult dosage is generally one tablet per day but this is often tailored to the needs of individual patients. In some cases, it is more appropriate to consume two dosages of 18.75 mg each on a daily basis. Phentermine is normally not suitable for pediatric patients, but this hasn’t stopped physicians from prescribing them to anyone over the age of 15.
Phentermine hydrochloride is similar to white crystalline powders. The substance is highly soluble in both water and alcohol. Some of the ingredients in the substance include starch, isopropyl alcohol, titanium dioxide, proplyene glycol, and ethyl alcohol.
The drug was commonly sold as Fastin, which was manufactured by King Pharmaceuticals, when it was first introduced to the market. Phentermine has been marketed under the names adipex, phentrol, sinpet, supremin, umine, panbesy, duromine, weltmine, and suprenza.
Phentermine hydrochloride has a long and illustrious history when compared to other weight loss drugs and substances. However, there have been very few if any clinical trials that have been carried out to understand the effects of phentermine hydrochloride on human beings. As phentermine alone has received very little heat from authorities, it has never been the focus of the FDA. However, the combinations of phentermine with fenfluramine and dexfenfluramine were extensively studied in 1996 and 1997 thanks to extensive negative press that the combinations, which were not FDA-approved, were getting. This would eventually lead to the removal of Fen-Phen and Dexfen-Phen from the market.
Fen-Phen and Dexfen-Phen
In the 1970s, phentermine hydrochloride was commonly used in tandem with other drugs to produce a powerful appetite suppressant. Specifically, it was prescribed by numerous physicians along with fenfluramine. Fenfluramine was approved as an anorectic agent by the FDA in 1973. This drug would become a part of the infamous Fen-Phen combination, which hit the shelves in the early 1990s. The Fen-Phen combination was marketed a wonder blend that was highly effective for fat burning and weight loss.
The FDA never legally approved this drug combo but that did not stop physicians from prescribing it. Numerous physicians benefited by selling the drugs together and this combination was regarded to be an effective, off-label medication.
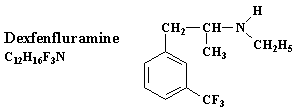
Phentermine was also used in tandem with dexfenfluramine, the Dexfen-Phen combination. Dexfenfluramine was approved by the FDA in 1996 as an appetite suppressant and its union with phentermine became an overnight sensation.
However, the rails began to fall off as this wonder combination was found to have induced negative side effects in its users. The Mayo Clinic findings of 1996-97 were a game changer for Fen-Phen and Dexfen-Phen. In 1997, the authors of a report published in the New England Journal of Medicine (NEJM) suggested a link between sustained use of these anorectic agents and mitral valve dysfunction, which is basically a heart valve disease. The paper estimated that in 1996 the Fen-Phen combination had been prescribed to nearly 20 million people. A 30-year-old woman, who had been using the Fen-Phen for a month, developed heart complications in 1996 and passed away shortly after. The Boston Herald published a front page article in her memory in the aftermath of her death in February 1997, lambasting the drugs responsible for her death.
Multiple patients filed grievance reports at this time, citing the ill effects of Dexfen-Phen and Fen-Phen on their bodies. People all over the country were reporting side effects on a daily basis, putting pressure on the FDA and American Home Products. Patients, who had consumed only fenfluramine and dexfenfluramine, without combining them with phentermine, were also reporting side effects. As these drugs were all FDA-approved, neither combination had ever been tested for safety by the FDA or American Home Products. The Mayo Clinic had reported twenty four separate cases of heart valve disease by the summer of 1997, all linked to the various combinations of phentermine.
The FDA could no longer sit idle and by September 1997, it requested drug manufacturers to withdraw fenfluramine and dexfenfluramine from the market. Dr. Michael Friedman, who was the FDA’s Lead Deputy Commissioner at the time, sent out a press release, publicly condemning fenfluramine and dexfenfluramine. The FDA recommended that consumers should consume neither of the drugs as nearly 30% of the users who had been a part of the Mayo Clinic report stated that they were experiencing abnormal heart valve issues. However, phentermine received a stay of execution and continued to be commercially available.
What exactly is phentermine and how does it work?
Phentermine is a substituted amphetamine based on the amphetamine structure. It is chemically and pharmacologically similar to the other types of drugs that have used as obesity treatment over the years. Phentermine and other drugs of this type are considered to be anorexigenics or anorectics. It is also pharmacologically similar to substances such as methamphetamine (crystal meth), MDMA, ephedrine, and amphetamine itself.
The substance is a psychostimulant drug and it possesses hallucinogenic properties. As all amphetamine-based substances are considered to be controlled substances, phentermine is classified as a Schedule IV controlled substance because it apparently possesses a high potential for abuse. The drug should always be taken with extreme precaution and users must never take multiple doses of phentermine as it can be as addictive as crystal methamphetamine and ecstasy.
The chemical name of phentermine is 1,1-Dimethyl-2-phenylethylamine. It is a synthetic organic compound. The chemical formula of phentermine is C10H15N. The molecular weight of synthesized phentermine is 149.23 g/mol.
Phentermine is a psychostimulant as it stimulates the central nervous system and elevates blood pressure. This is how it functions as an appetite suppressant. Phentermine is a TAAR1 agonist as it activates TAAR1 in monoamine neurons, thereby facilitating the release of these neurons into the synapse. Phentermine primarily releases norepinephrine from neurons and dopamine & serotonin to a lesser extent.
Phentermine is a substance that causes neurons to expunge or maintain levels of a group of neurotransmitters that are called catecholamines. Dopamine and norepinephrine are a part of this group. The drug appears to inhibit reuptake of these neurotransmitters by inhibiting or reversing the reuptake transporters. The drug also inhibits MAO enzymes, which causes a greater amount of neurotransmitters to remain at the synapse.
By elevating these catecholamines, experts hypothesize that the drug may have an impact on the brain’s leptin levels. Phentermine is believed to raise the brain’s leptin levels. The elevation of catecholamines is also believed to halt the chemical messenger neuropeptide Y, a peptide responsible for increasing hunger and fat storage.

Phentermine primarily works to reduce obesity by reducing the body’s hunger perception. This is mainly done through a cognitive process by working on several nuclei in the hypothalamus section of the brain. The drug also works outside the brain by releasing adrenaline and noradrenaline to ensure that fat cells break down stored fat in the body.
Usage and administration
Phentermine consumption must always be accompanied by weight reduction-based exercises, reduced intake of calories, and suitable behavioral modification, all of which must be based on an individual’s level of weight. Ideally, phentermine should be consumed in the morning an hour or two before the first meal of the day. Never consume phentermine in the evening or especially after a meal. Patients are advised to not consume phentermine in the evening due to the possibility of insomnia.
Phentermine is normally available orally. It is available in orally dissolving capsules and tablets in a wide range of strengths, often based on the brand. Consumption of phentermine by any other form is ill advised. Suprenza is an example of an orally disintegrating form of phentermine. Patients who consume suprenza must allow the drug to simply dissolve on their tongues.
Different brands or labels of phentermine have different administration periods. Most brands require you to take the drug before breakfast on an empty stomach, while there are some brands that recommend consumption between breakfast and lunch. Don’t follow the instructions on the label and consult your doctor for the most accurate instructions based on your medical history.
Never consume phentermine or any of its combinations in tandem with alcohol as the side effects of the drug is likely be extra harmful when the body is forced to break down alcohol and phentermine together. You should avoid any medication that has the potential to dull the nervous system, like alcohol does, when taking phentermine. As phentermine stimulates the nervous system and alcohol inhibits it, the two work to cancel each other out and prevent the desired effects of phentermine from bearing fruit. This is in fact a bigger problem with alcohol consumption than any possible side effects.
If you are being administered monoamine oxidase inhibitors, do not, under any circumstances, consume phentermine concurrently. Patients should wait for a period of 14 days after completing their consumption of monoamine oxidase inhibitors to begin their consumption of phentermine.

If you find yourself developing a tolerance for your phentermine medication, do not start exceeding your recommended dosage. Instead, you should stop consuming phentermine and ask your physician to prescribe another weight loss drug. Alternatively, you can stop using phentermine for a couple of months which should remove your tolerance for the anorectic.
Despite the fact that phentermine in its current forms has no minimum administration period, the medication should ideally not be taken for more than 12-16 weeks at a stretch as it can lead to serious long-term consequences if it’s present in the body for a lengthy period of time. When you are using phentermine, make sure that you store the drug at room temperature. Keeping it away from heat and moisture is of utmost importance.
People who engage in potentially dangerous activities on a daily basis such as operating heavy machinery must take extra caution. As phentermine has the ability to mess with the body’s motor functions and disrupt heartbeat, such patients must take care to followed a personalized, physician-approved plan before consuming phentermine on a regular basis.
If you are suffering from diabetes mellitus and taking medication, it is advisable to reduce the level of your oral hypoglycemic or insulin medication in a controlled manner before starting your course of phentermine.
Experts specify that certain types of people should not be prescribed phentermine because of its contraindication properties. Specifically, its use can be contraindicated in people who have glaucoma, a history of drug abuse, hyperthyroidism, a history of cardiovascular disease, and hypersensitivity. Individuals who are currently taking another form of amphetamine medicine such as adderall are also advised against consuming phentermine.
Pregnant women, women who are planning on becoming pregnant or those who are breast-feeding are also discouraged from using phentermine. This is because phentermine will pass through the breast milk of a woman who is breast feeding and directly to her baby. The effects of phentermine on small children are unknown as there have been almost no instances of such cases, but it is highly reasonable to assume that it will not end well for babies.
Why has phentermine been historically popular?
According to IMS health, a body that monitors the use of prescription drugs, phentermine commands 80% of the weight loss and diet drug market as of 2015. This is remarkable for a drug that has had huge question marks about its side effects on the human body. So how exactly has it always been so popular?
Phentermine and phentermine hydrochloride gained popularity as being short-term cures to obesity. In the twentieth century, phentermine was one of the very few weight loss drugs available in the market. The price of phentermine has always been low when compared to its competitors, some of them that have way fewer side effects. Other diet drugs such as Xenical and Meridia are priced significantly higher than phentermine. Nowadays phentermine is often $30 for an entire month’s supply, a price that blows its competition out of the water.
There have been numerous allegations over the last fifty years that physicians have directly and indirectly benefited from prescribing phentermine and its various combinations to money conscious patients. However, this is nothing out of the ordinary when it comes to prescription drugs as pharmaceutical companies have time and again been accused of these practices.

Phentermine’s popularity is also historically linked with the popularity of fenfluramine. When fenfluramine was developed in the 1970s, the drug was expected to become a market leader in the segment. However, this was vastly different from the ground reality. Most patients who consumed fenfluramine complained of effects such as anxiety and nausea on a large scale basis. American Home Products manufactured the drug under the name Pondimin, but the market sales were extremely poor and it never caught on as a suitable alternative to regular exercise and dieting.
In 1992, researchers at the University of Rochestor stated that the combination of phentermine and fenfluramine was incredibly effective as an obesity combatant. The research team, led by Dr. Micheal Weintraub, believed that this combination did not cause the same irritating side effects that other diet drugs did. This led to a huge surge in the popularity and sales of phentermine. Fen-Phen was highly successful in the market as there were cases in which patients were able to drop as much as 30 pounds in a couple of months.
As the popularity of Fen-Phen soared, American Home Products was worried as the patent the company held for Pondimin was about to expire. The company was against the idea of its competitors getting their hands on fenfluramine and the burgeoning profits of Fen-Phen. American Home Products had to find a solution to this problem before their market share dipped.
This problem was solved by the introduction of Redux to the market. American Home Products and its subsidiaries developed a brand new version of fenfluramine, which happened to be known as dexfenfluramine. The FDA was particularly wary about this new weight loss drug, but it approved the drug in 1996 after intensive debates about its potentially harmful effects.
The introduction of Dexfen-Phen reinvigorated the market, and it was thought to be even better than Fen-Phen when it came to fighting obesity. Just like the fenfluramine and phentermine combine, Dexfen-Phen was not intensively tested for safety. The company’s focus at this time was to ensure that the FDA would approve its new product before the patent on fenfluramine ran out. As such, it was not highly preoccupied with the adverse effects of dexfenfluramine on the people who consumed it.
As the sales of Dexfen-Phen were rising in 1996, multiple reports of its adverse effects and its links to coronary heart disease surfaced and a few patients consuming the medication ended up dead, including some patients who had a history of clean health. The FDA could no longer sit idle and it banned the commercial distribution of dexfenfluramine and fenfluramine.
What are the side effects of phentermine?
Phentermine has multiple side effects that have come to the fore in various ways over the years. Phentermine’s contentious history has time and again led to numerous experts to call for its removal from the market, but the drug has managed to stay in distribution thanks to a number of factors.
Some of the side effects of phentermine include severe chest pain, decreased energy levels, dizziness and fainting, irregular and rapid heartbeat, swelling and trembling of the feet, thought impairment, headaches, and numbness of the arms and legs. The effects also include restlessness, euphoria, hypertension, diarrhea, dry mouth, facial oedema, impotence, urticaria, and arrhythmia.

Phentermine has been known to have various gastrointestinal effects on the body. This primarily includes vomiting and cramps. Acute transient ischaemic colitis has also been linked to regular intake of phentermine. The dermatological effects of the drug include pale skin and drying of the mucous membrane. The drug also has notable genitourinary effects. Acute urinary retention is a common symptom. Renal ischemia is possible, and renal failure can also occur in tandem with dehydration and rhabdomolysis.
Phentermine is also known to have severe effects on the body’s endocrine system. Patients who have been consuming phentermine for at least eight weeks have been known to report significant changes in libido in addition to instances of decreased sexual interest and impotence.
Geriatric patients have also been known to be more sensitive to phentermine. Geriatric patients should be prescribed very small amounts of phentermine to begin with. This is because their cardiovascular, renal, endocrine, hepatic, and other functions are likely to be much lower than that of younger adults. The kidneys are known to excrete substantial amounts of phentermine and this is more likely to lead to toxic reactions in geriatric patients, as they are likely to have weaker renal functions.
There are numerous medications that may interact with phentermine and produce harmful side effects. Dexfenfluramine and fenfluramine are primary examples of this problem, having played a great role in causing heart diseases. Substances such as phenelzine and furazolidone are contraindicated because they may lead to elevated blood pressure levels, decreased heart rate, elevated body temperature levels, and even severe lung problems.
Potential to cause heart disease and pulmonary hypertension

Cardiovascular diseases associated with phentermine have been suspected since the 1970s. Various causes of palpitations, ischemic events, and tachycardia have been reported. Although a large majority of these cases have been reported by people taking the Fen-Phen or Dexfen-Phen combo, rare cases of patients who have only taken phentermine have reported valvular heart disease and primary pulmonary hypertension. People who have been shown to be healthy before being administered phentermine have developed heart diseases where no other substance could have possibly led to the defect, barring inherent genetic defects.
Fen-Phen and Dexfen-Phen have been also theorized to lead to primary pulmonary hypertension (PPH). PPH is a rare, but fatal disease of the lungs. However, just as with cardiovascular diseases, the possibility of phentermine alone leading to PPH cannot be discounted. There have been rare cases of people reporting PPH, having consumed phentermine alone. It is not clearly understood if these people had any underlying genetic issues that could have led to PPH.
Dyspnea, which refers to labored or difficult breathing, is typically the initial symptom of PPH. Individuals who experience any breathing difficulties or exercise intolerance as a result of consuming phentermine are advised to consult their physician without any delay. If other symptoms such as lower extremity edema and angina pectoris show up alongside dyspnea, patients should immediately stop their consumption of phentermine and evaluate themselves for possible signs of primary pulmonary hypertension.
Current forms of phentermine
Currently one of the most popular variations of phentermine is Qsymia. Qsymia is a combination of topiramate and phentermine. This is a part of recent efforts to study phentermine in combination with other drugs to increase its effectiveness as a weight loss supplement and reduce its negative side effects. Qysmia was approved by the FDA for public consumption in 2012.
For adults who are starting out on Qysmia, the dosage should be no more than phentermine 3.75 mg/topiramate 23 mg for the first two-three weeks of consumption. After this initial period, you can normally double our dosage but this general rule does not apply to everyone. Qysmia has no minimum administration period, but if you do not experience at least 3-5% weight loss after consuming Qysmia for three months, consult your doctor as the drug is unlikely to help you achieve the desired results.
Qysmia carries around a lot of the same health risks that characterized earlier forms of phentermine. It remains to be seen if Qysmia has any similar long-term effects on the body that Fen-Phen and Dexfen-Phen had. As of now, no relationship between Qysmia and mortality or cardiovascular morbidity has been established.
Knowing the patient’s body mass index before administering Qysmia is of the utmost importance. Qysmia is normally prescribed to adults who have an initial body mass index (BMI) of 30 kg/m2 or greater. People above this BMI are considered to be clinically obese. It is also suitable to patients who have a BMI of 27 kg/m2, which is considered to be overweight. These patients must be suffering from a weight-related comorbidity such as type two diabetes or hypertension.
Patients who are consuming Qysmia are advised against discontinuing the drug abruptly. If you want to stop taking Qysmia, you need to do it in a gradual fashion. Take a dose of Qysmia every other day for at least one or two weeks before discontinuation. This is advised as abrupt discontinuation has been known to lead to violent seizures for both Qysmia and other forms of phentermine.
Qysmia should not be consumed by pregnant women and women attempting to become pregnant. Weight loss during pregnancy and the presence of Qysmia in the bloodstream can be detrimental to new born children. Women are advised to use strict birth control measures during Qysmia intake to avoid pregnancy. It is suitable to make sure that you are not pregnant before starting a course of Qysmia medication.
Another current popular form of phentermine is the Phen-Pro cocktail. Pro stands for prozac. Substances containing prozac that are commonly used in tandem with phentermine are Effexor, Zoloft, Luvox, and Celexa. This combination of prozac and phentermine is considered to an off-label combination, just like Fen-Phen and Dexfen-Phen. This means that the combination is not FDA-approved, and there may be adverse side effects to this combination that are currently not understood by the FDA and the companies producing the drugs. So you should ideally avoid this combination, considering the devastating effects of the Fen-Phen and Dexfen-Phen combinations.
Always consult your doctor and have a comprehensive knowledge of your medical history before consuming phentermine. It is not a drug to be taken lightly. It may potentially give you excellent results in the fight against weight loss, but it could also very easily have an adverse impact on your health. If you are not comfortable with phentermine, there are several, albeit more expensive, alternatives in the market. Consult a trustworthy doctor and create a personalized plan for yourself.



















